Abstract
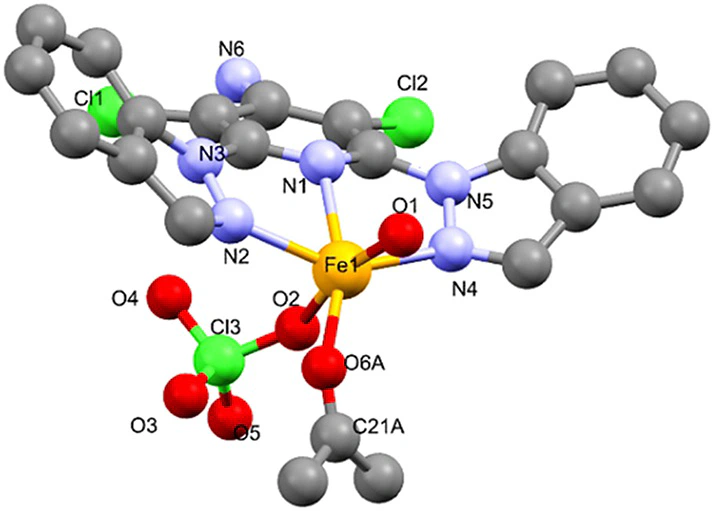
4-(2,6-Di(2H-indazol-2-yl)pyridin-4-yl)benzoic acid (1) and 10-(2,6-di(1H-pyrazol-1-yl)pyridin-4-yl)anthracene-9-carboxylic acid (2) were required for adsorption studies on Ag(111), with a view to subsequent iron(II) complexation and formation of well-ordered spin-responsive self-assembled monolayers. While the generation of these compounds has remained elusive, several intermediates and by-products were obtained, potentially useful as dipyrazolylpyridine-related derivatives and for metal ion coordination. 3,5-Dichloro-2,6-diindazolylpyridine-4-amine, which forms as a mixture of regioisomers, was synthesised, the mixture separated, and the components characterised (3,5-dichloro-2,6-di(2H-indazol-2-yl)pyridin-4-amine; 3,5-dichloro-2-(1H-indazol-1-yl)-6-(2H-indazol-2-yl)pyridin-4-amine; 3,5-dichloro-2,6-di(1H-indazol-1-yl)pyridin-4-amine). Their iron(II) complexes have been prepared and fully characterised, including single crystal X-ray structure determination. The complexes are instructive examples of the influence of ligand design (“steric jamming”) on the spin-crossover (SCO) activity of FeII centres. Bulky substitution, which entails twisted ligand conformation, increases intramolecular crowding. This prevents contraction of the metal coordination sphere, which would be a prerequisite for thermally inducible SCO. Mössbauer spectroscopy has revealed that the complexes remain predominantly high-spin (HS) between 20 and 200 K, and that a mixture of conformational HS isomers is present in the microcrystalline solid.