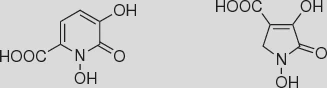
„… und ehrt mir ihre Kunst!” – Evaluierung historischer und neuer Synthesewege zu 1,5-Dihydroxy-6-oxo-1,6-dihydropyridin-2-carbonsäure und 1,3-Dihydroxy-2-oxo-3-pyrrolin-4-carbonsäure
Abstract
The historical and recent literature describing synthetical pathways to the title compounds is comprehensively summarized and evaluated. Routes via 2,5-dioxoadipic acid or 2,5-dihydroxymuconic acid as intermediates could not be followed reproducibly. 3-Hydroxy-2-oxo-2H-pyran-6-carboxylic acid and ethyl 3-hydroxy-2-oxo-3-pyrroline-4-carboxylate were fully characterized by spectroscopic methods and single-crystal X-ray diffraction. Both compounds have an enolic structure in solution as well as in the solid state. A recently published ring-closing metathesis (RCM) protocol to a derivative of the first title compound was unproductive in our hands. We present a new two-step synthesis of the similarly substituted 5-hydroxy-6-oxo-1,6-dihydropyridine-2-carboxamide using succinyl cyanide. The product was identified and characterized by spectroscopic methods and X-ray crystallography.